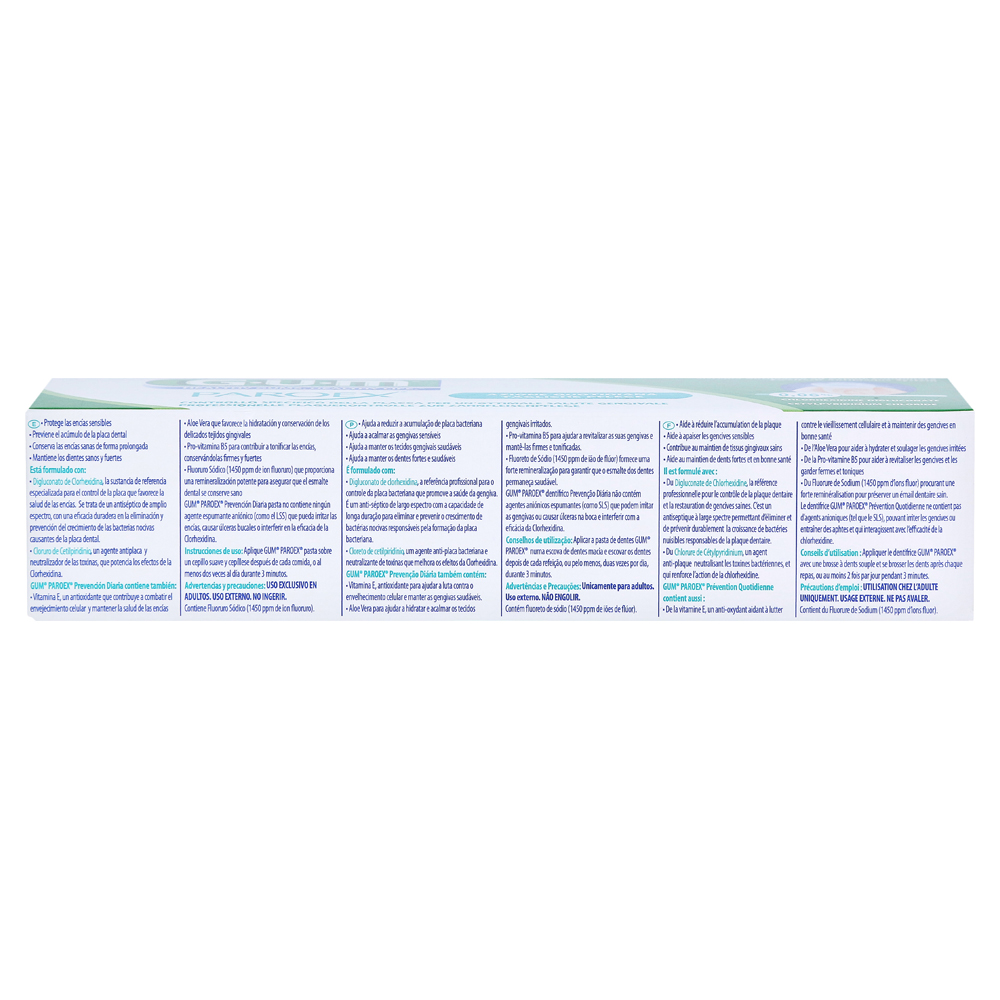
Sunstar Americas Inc. Expands Voluntary Nationwide Recall of Paroex® Chlorhexidine Gluconate Oral Rinse USP, 0.12% Due to Microbial Contamination | FDA

GUM Paroex (anti-gingivitis oral rinse) being recalled due to microbial contamination that could lead to respiratory and other infections in patients - Canada.ca

🦷 September is National Oral Health Month 🦷 I am truly obsessed with oral care, and these are 5 top-quality GUM oral care tools to help… | Instagram

Sunstar Americas, Inc. Issues Voluntary Nationwide Recall of Paroex® Chlorhexidine Gluconate Oral Rinse USP, 0.12%, 473ml due to Microbial Contamination

🦷 September is National Oral Health Month 🦷 I am truly obsessed with oral care, and these are 5 top-quality GUM oral care tools to help… | Instagram

Advisory - GUM Paroex (anti-gingivitis oral rinse) being recalled due to microbial contamination that could lead to respiratory and other infections in patients










/201412031203233n1rxcc552auapjf3fo3ehe2e.jpg)


:sharpen(level=0):output(format=png)/up/dt/2018/12/Fig-1A.png)



:sharpen(level=0):output(format=png)/up/dt/2018/12/Fig.-4B.png)











:sharpen(level=0):output(format=png)/up/dt/2018/12/Fig-1B.png)




:sharpen(level=0):output(format=jpeg)/up/dt/2018/12/Gum.jpg)